Biological Responses to Electric Fields and Non-thermal Plasma
The application of nanosecond pulsed electric fields of extreme field strengths has had amazing success when used on tumors and in accelerating wound healing. Melanoma
tumors could be send into complete remission and wounds treated platelet gel that was activated by nanosecond pulses closed roughly twice as fast as the untreated
controls. Apart from these imminent medical therapies, a variety of cellular and microbiological responses have been observed, including enhanced proliferation and
up- and down-regulation of genes.
Although the biological response to a nanosecond pulse electric field exposure shows on time scales long compared to the exposure, it is surely caused by the physical processes during the stimulus. In the first place, the strong electric field leads to a 'redistribution' of charges - ions and dipoles alike - along and within cellular structures. These are primarily charging mechanisms of cell membrane and internal membranes. In our understanding, this will eventually affect the structure of the membrane itself and even of macromolecules, such as DNA and proteins, when a sufficiently high charging voltage can be reached. As a consequence, the understanding of charging mechanisms and of the thresholds that can be associated with different subsequent biochemical mechanisms is obviously very important. Unfortunately our knowledge of the interaction so far mostly has to rely on simulation and modeling studies, which are 'ridden' by more or less accurate assumptions. Or in other words, measurements on charging processes with a resolution that is short compared to the already extremely short time of the exposure. (Remember, we are dealing with nanosecond exposures, i.e. even light will travel in 1 nanosecond only a distance of distance of 30 centimeter, i.e. about 1 foot!) We took on the challenge to develop fast diagnostic method for the investigation of cellular responses during (and before and after) the application of a nanosecond pulse electric field. One very unique method (to our knowledge the only one in the world) allows us to measure changes in the transmembrane voltage with a resolution of only 5 nanoseconds. The technique combines a stroboscopic illumination with a sensitive camera and an extremely fast voltage sensitive dye. (Details can be found in our publications.) Our experiments showed that only values on the order of 1.5 volts will be reached, which is much lower than predictions from some modeling studies that were on the order of 40 volts. Our experiments further show that this peak value is followed immediately by a 'slow' but steady decrease of the transmembrane voltage, even while the electric field is still applied.
We believe that these voltages will eventually lead to the permeabilization of membranes. The phenomenon is known for the application of much longer pulses of
microseconds and milliseconds (although of much lower field strength) as electroporation. However, hypothetical pores seem to be too small as to allow the
passage of large molecules. This is what makes 'electroporation' an appealing technology for gene therapies and drug delivery. For nanosecond pulse exposure initially
only small ions seem to move across the membranes. An indication is the slow decrease of transmembrane voltages already during the exposure, which is explained by
equilibration of the ion gradients in the applied field.
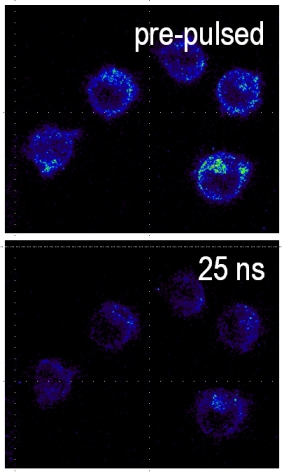
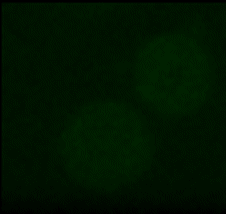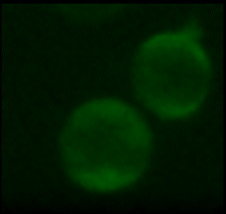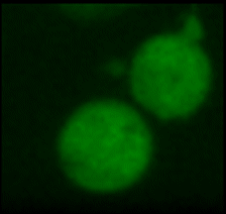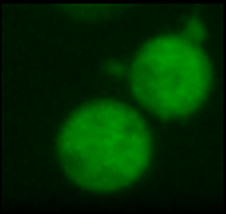
A very important ion that participates in this process is calcium. It is stored in different compartments in the cell and a release from these stores, or in general an increase in intracellular calcium levels can trigger a variety of cell specific responses, such as the aggregation of platelets (one of the first steps in the wound healing process). Changes in intracellular calcium levels have also been the fastest 'biochemical' responses that have been observed so far for nanosecond pulses. In general they are reported as occurring "as fast as you can look at them". We have managed a more quantitative analysis and found that calcium levels will rise within a few milliseconds after the application. We are further, fairly certain that the increase is caused by a permeabilization of the membrane of the endoplasmic reticulum. What is probably even more interesting that how much the calcium level changes depends on the strength and duration of the applied field. This offers possibilities to actually 'regulate' intracellular calcium levels and instigate specific responses.
Our efforts on the immediate biophysical effects of pulsed electric fields are ongoing. What we currently really would like to know is, how intracellular membranes
charge but also how tissues are charging in comparison to a single cell. In addition we are trying to understand how the electric field actually interacts with the
constituents of membranes on a molecular level and how this will lead to the formation of pores. Beyond that immediate goal, we are further trying to figure out, if
we cannot actually affect molecules, such as the DNA, directly and with a lasting effect by a nanosecond pulsed electric field. There's still much to do.
For more details or information check out our publications or send me an email.
|
 |
Projects Coming soon... (hopefully) |